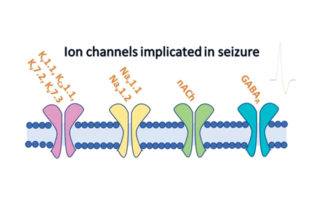
An Integrated Approach for Early In Vitro Seizure Prediction Utilising hiPSC Neurons and Human Ion Channel Assays
An Integrated Approach for Early In Vitro Seizure Prediction Utilising hiPSC Neurons and Human Ion Channel Assays ApconiX are delighted with our recent publication in Toxicological Sciences where we presented our new data on our integrated approach to detecting seizure. After 3 years of research, we launched the seizure [...]