Improving Sustainability in Our Ion Channel Laboratory
Introduction
Hannah Jennings presented a poster at ELRIG Drug Discovery in London earlier this year. Her work looked at reducing plastic waste in the laboratory by using assay-ready cells for ion channel work. This was very successful, contributing to a reduction in our average laboratory turnaround time to 3.9 days. Although this is an average and varies depending on ion channel tested, we are very pleased that sustainability can have such a positive impact on the service we provide our clients.
Bethan Kelly, our industry placement student, is carrying out a project to reduce the use of fetal bovine serum (FBS) in cell culture. This would make our ion channel laboratory completely animal-product-free.
Both projects have helped the laboratory reduce consumables, which in part has helped offset some of the higher costs that are currently being felt everywhere.
Here they describe the work of both projects.
Making Sustainability an Integral Part of Everyday Lab Work
As an environmentally conscious company, we are always looking for ways to make sustainability an integral part of everyday lab work and to reduce plastic waste where we can. Each year the consumables and equipment required for day-to-day cell culture generate excessive plastic waste, which cannot be reused or recycled. Work in ion channel screening means that our lab relies heavily on single-use plastics in day-to-day operations.
It was estimated that up until 2022, each month our cell culture department would generate an average of 80kg of consumables and plastic waste, most of which cannot be recycled due to contamination.
Assay Ready Cells
The use of assay-ready cells in place of continued cell culture has been known to save time and reduce the amount of plastic waste generated, compared with traditional cell culture methods. These are cells frozen at high viability, making them fit for drug safety screening, and can be used in patch-clamp assays directly after thawing without culture or passage.
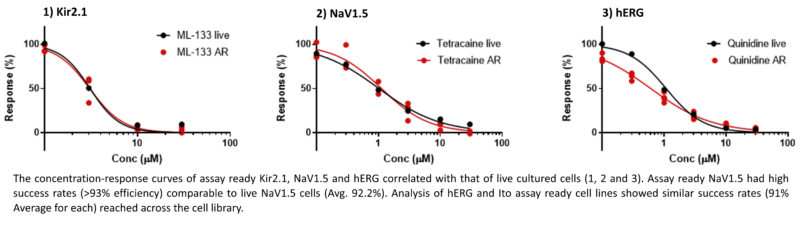
Over the last 6 months, we have conducted a study where we generated assay-ready cell lines expressing NaV1.5, Ito, hERG and Kir2.1 and tested their performance on automated patch-clamp platforms. These cells were frozen and stored at high density and thawed immediately before assay, eliminating the need for prolonged culture. The assay-ready cells have shown excellent viability and functional activity comparable to cultured cells as determined by the similarity of concentration-response curves.
Reducing Turnaround Time
Additionally, this method has allowed us to streamline laboratory processes by saving time spent in cell culture, thus improving laboratory efficiency. In 2022, around 80% of hERG studies were reported in one week or less. Our average turnaround time was 3.9 business days from compound receipt to reporting. These steps have also minimised the amount of plastic and consumable waste generated from cell culture by ~50% over the last 6 months.
Following the success of this initial study we are pleased to say that expanded testing is ongoing to confirm reproducibility of highly functional assay ready cell lines, and to implement these methods across all cell lines, especially those for our CiPA panel, in use at ApconiX.
Reducing the Use of Fetal Bovine Serum
The success of the study has since inspired work on reducing the use of fetal bovine serum (FBS) in cell culture. FBS is a widely used component of cell culture media as it contains high amounts of nutrients and growth factors to support cell growth and proliferation. However, FBS raises ethical concerns. The lab is interested in making the change to serum-free media; this change will also address batch-to-batch variation providing the consistency needed for optimum patch clamping.
Since FBS is complex and undefined, a few weeks will be spent adapting the hERG cell line to serum-free media conditions and determining what supplements are required to maintain cell viability and protein expression. If successful, the same efforts will be applied to other cells lines used by ApconiX with the expectation that a serum-free and chemically defined media alternative can be developed. This will make the lab animal-product free, another step in the right direction towards sustainability.





