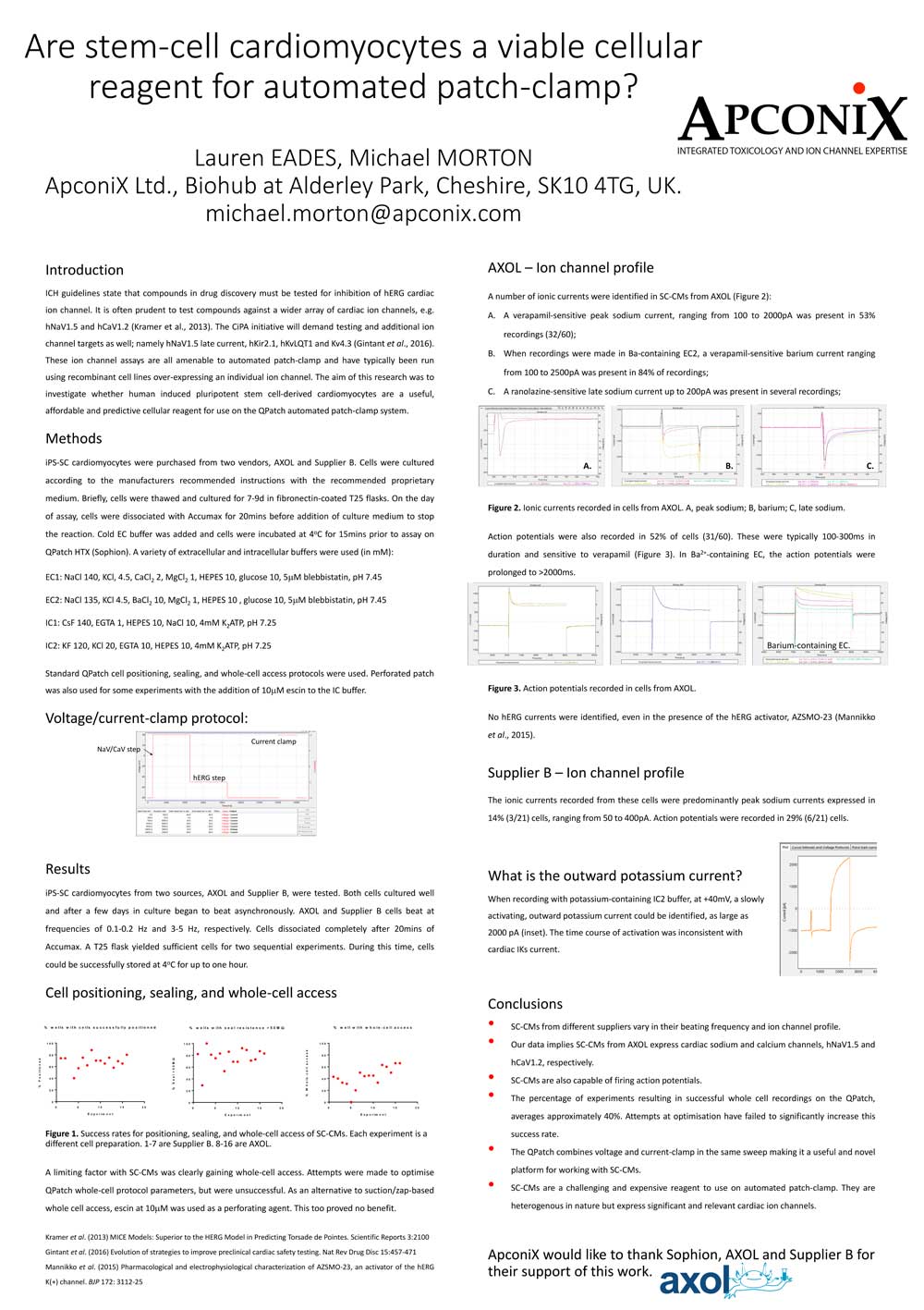
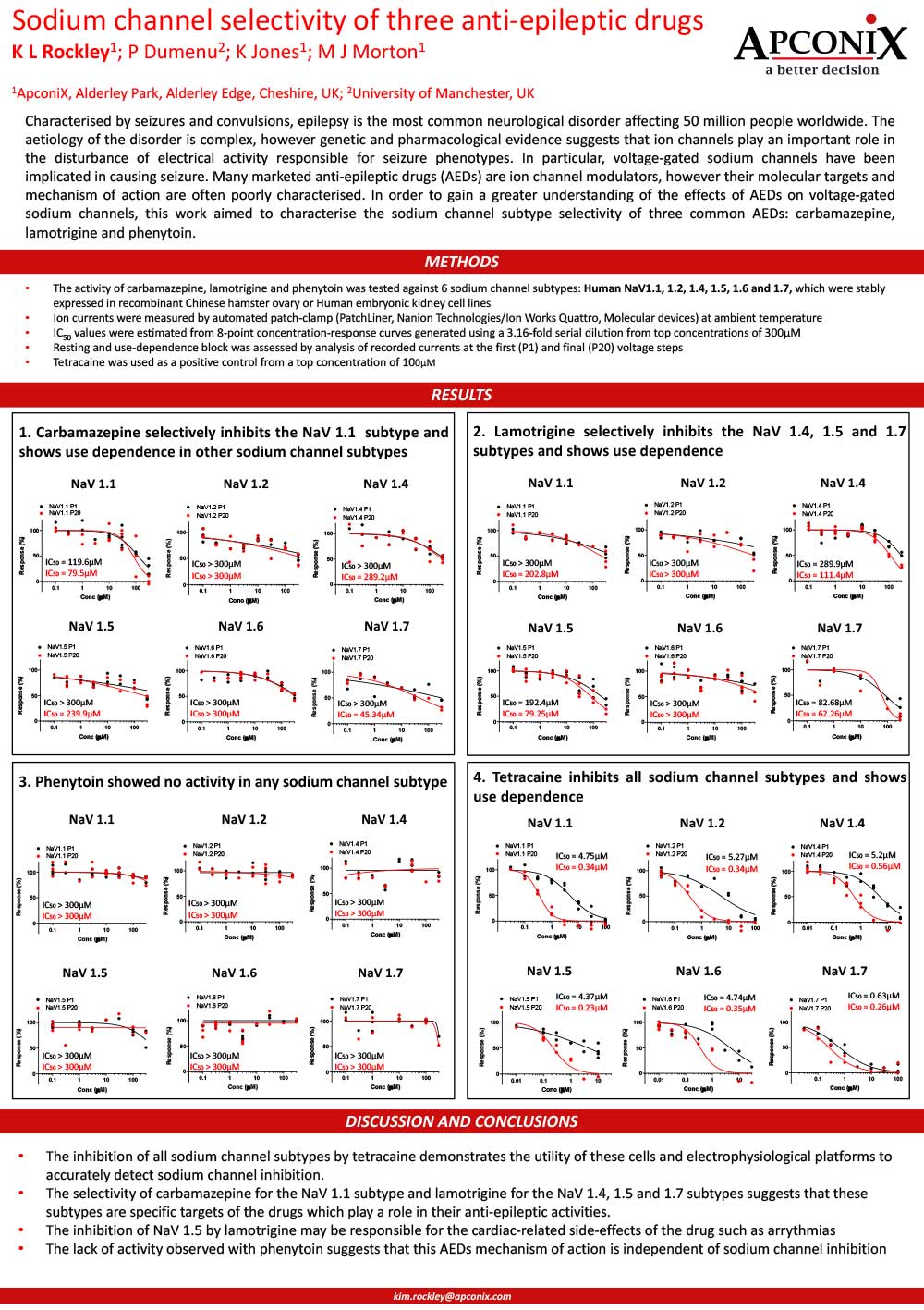
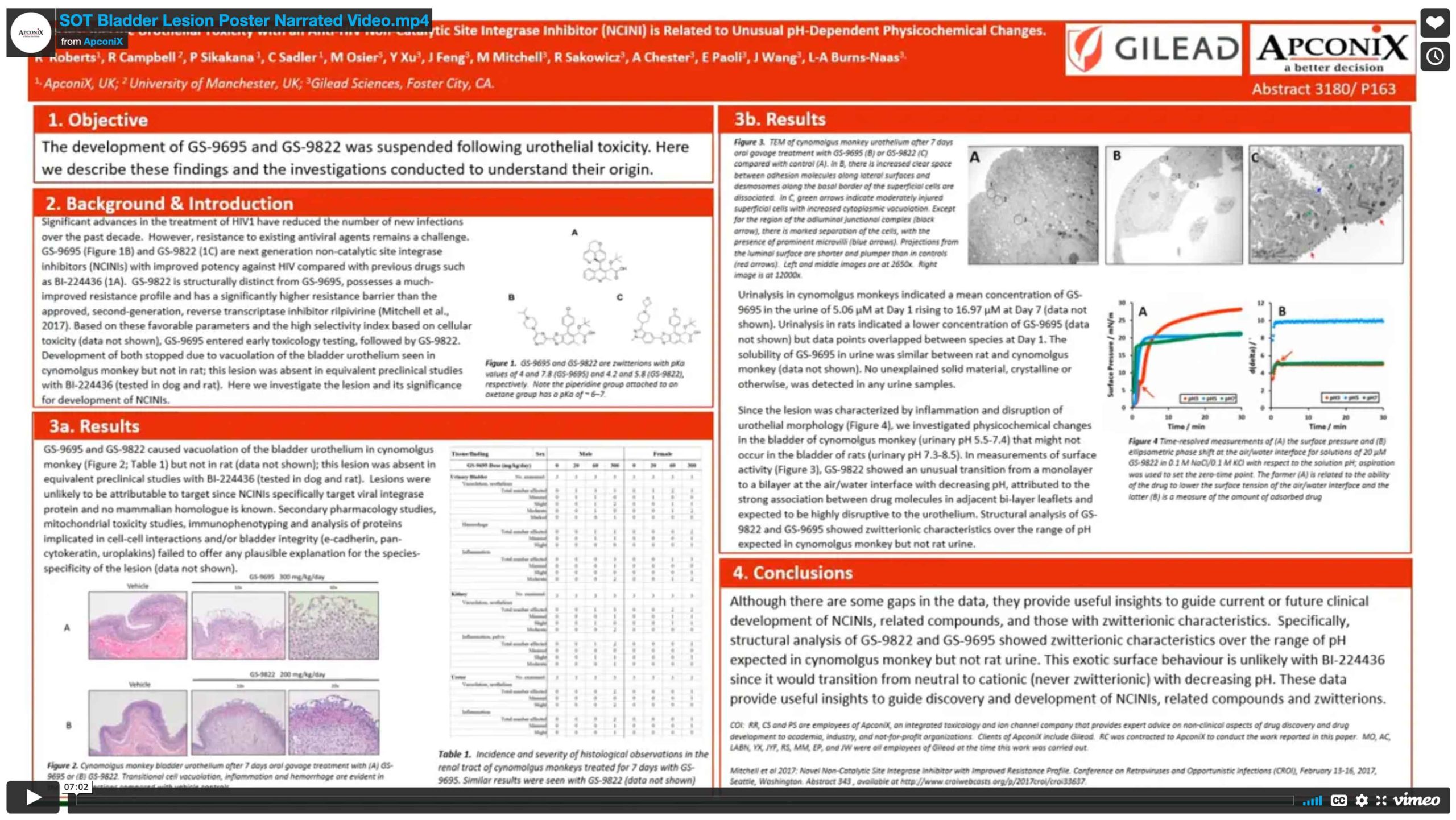
Download our posters resources here
Welcome to Apconix’s Resources page – click on the button below to download the resource.
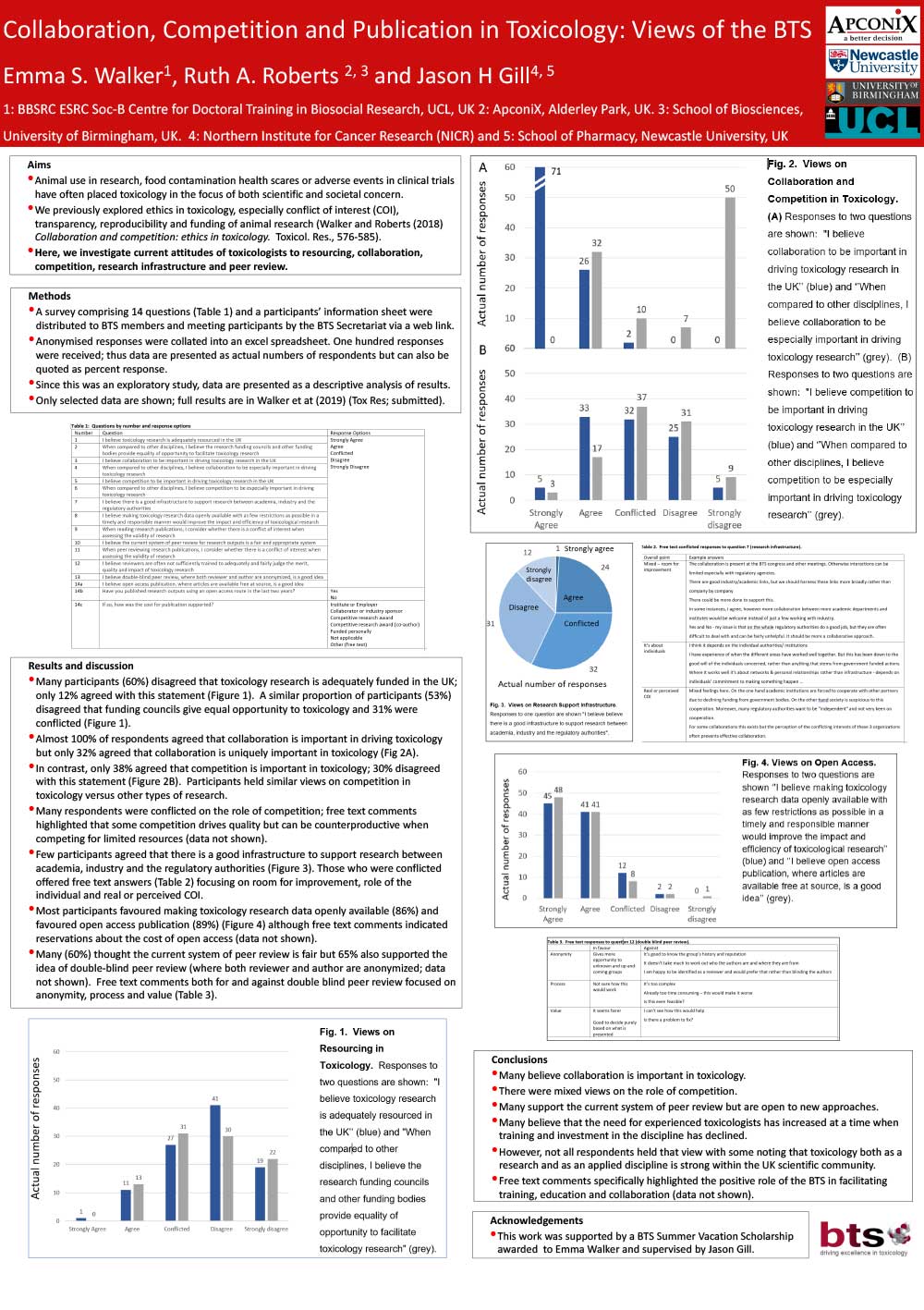
Collaboration, Competition and Publication in Toxicology Views of the BTS
[sdm_download id="10343" fancy="0"]
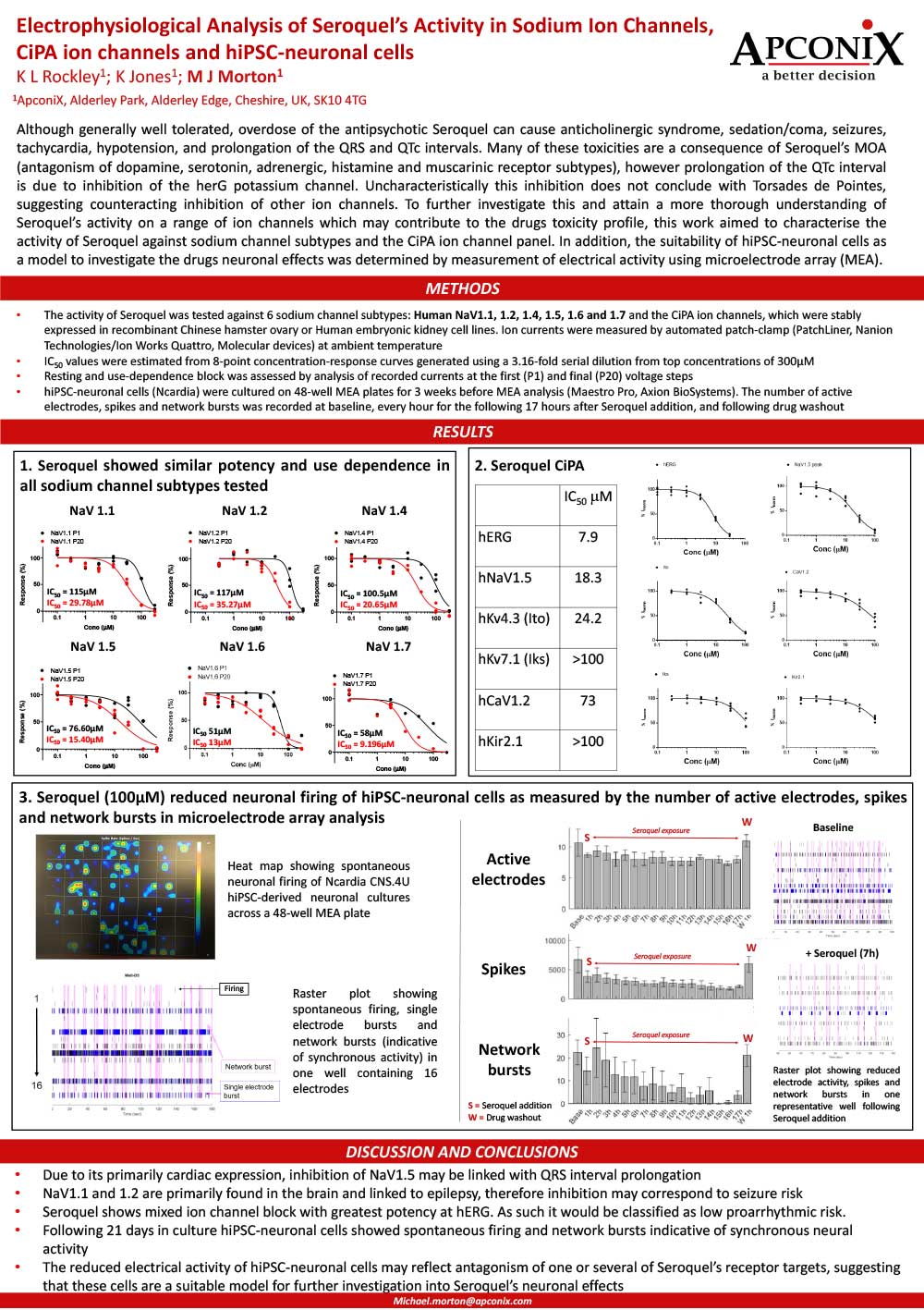
Electrophysiological Analysis of Seroquel’s Activity in Sodium Ion Channels CiPAion channels and hiPSC neuronal cells
[sdm_download id="10346" fancy="0"]
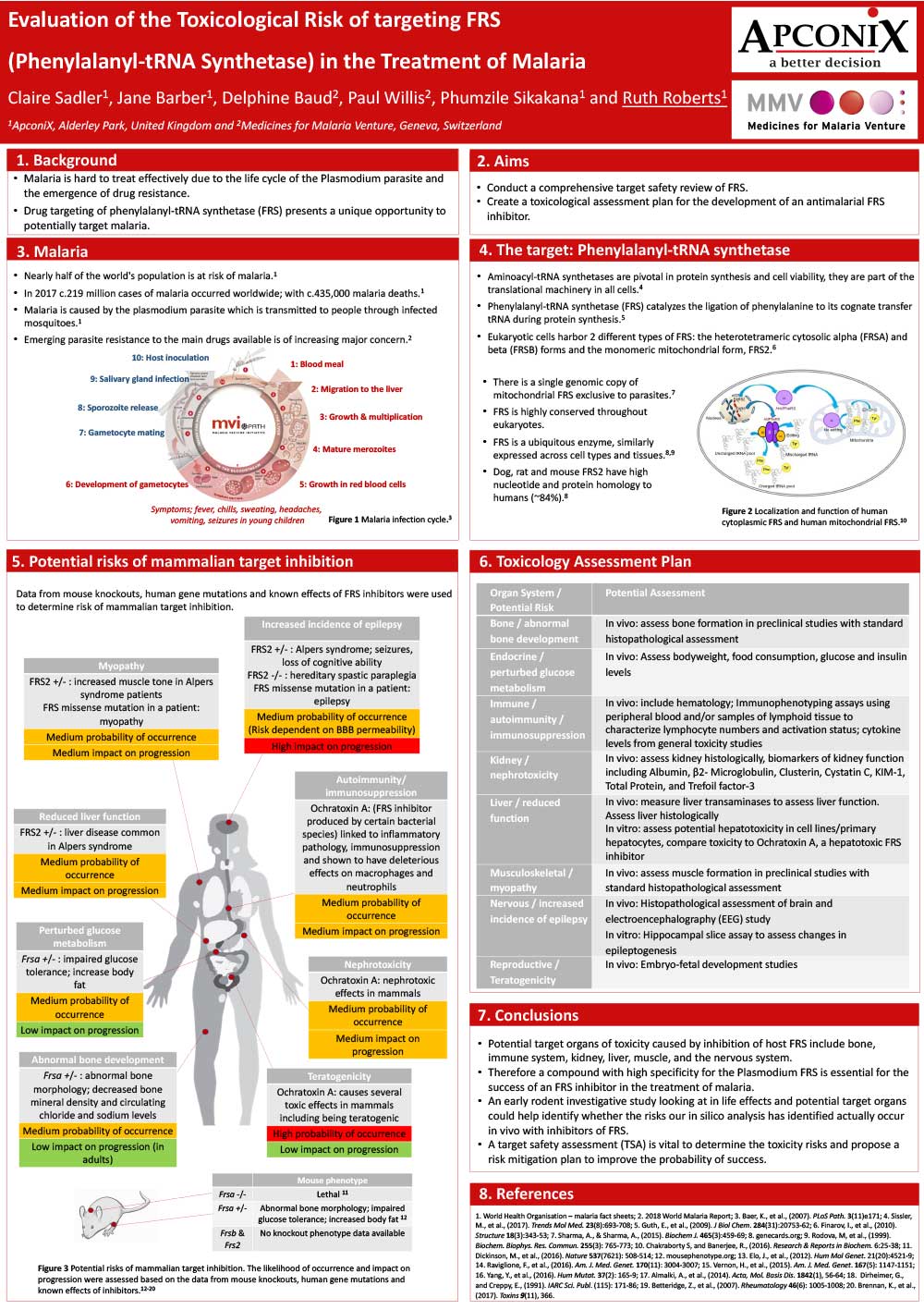
Evaluation of the Toxicological Risk of targeting FRS (Phenylalanyl-tRNA Synthetase) in the Treatment of Malaria
[sdm_download id="10345" fancy="0"]
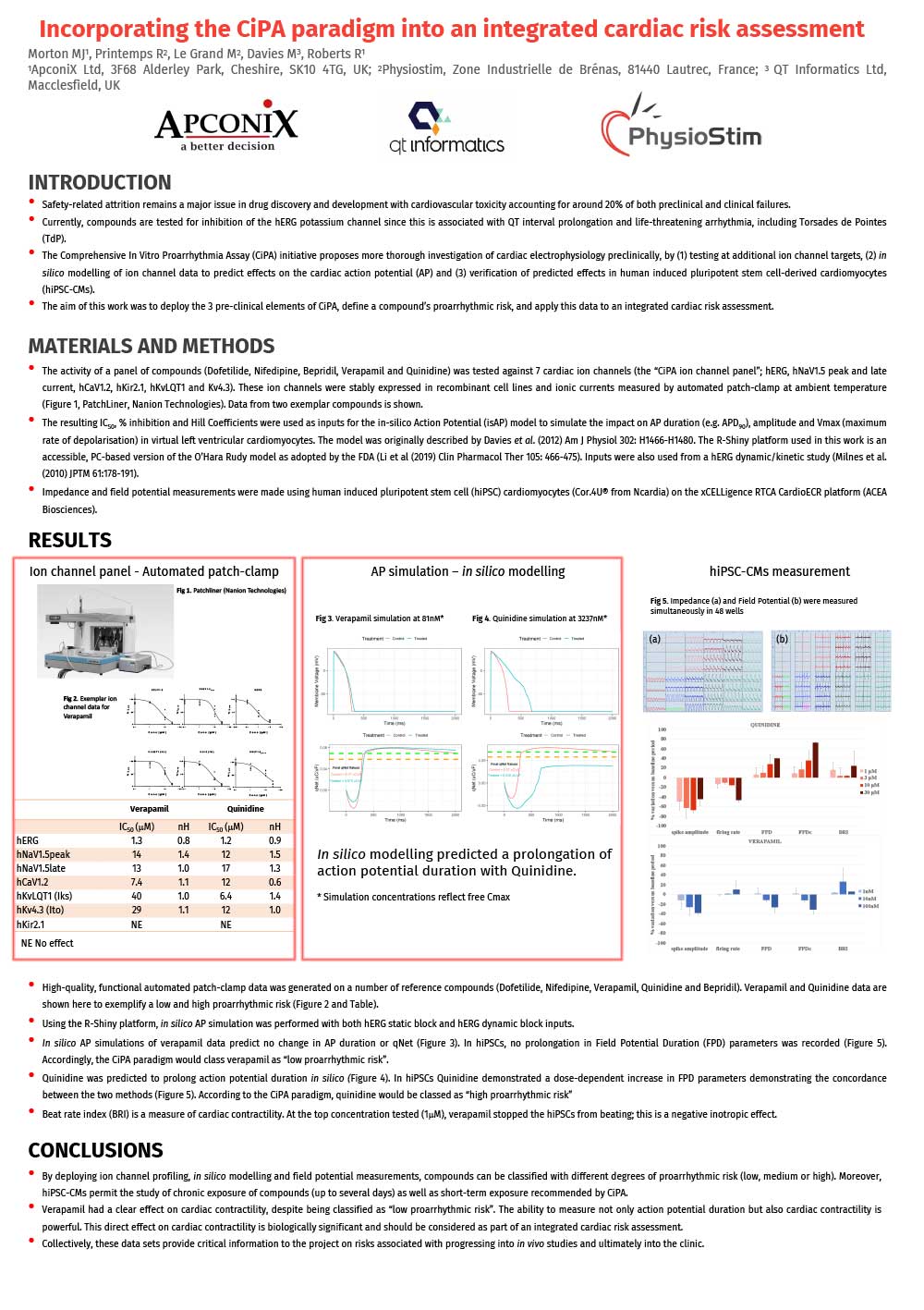
Incorporating the CiPA paradigm into an integrated cardiac risk assessment
[sdm_download id="10347" fancy="0"]
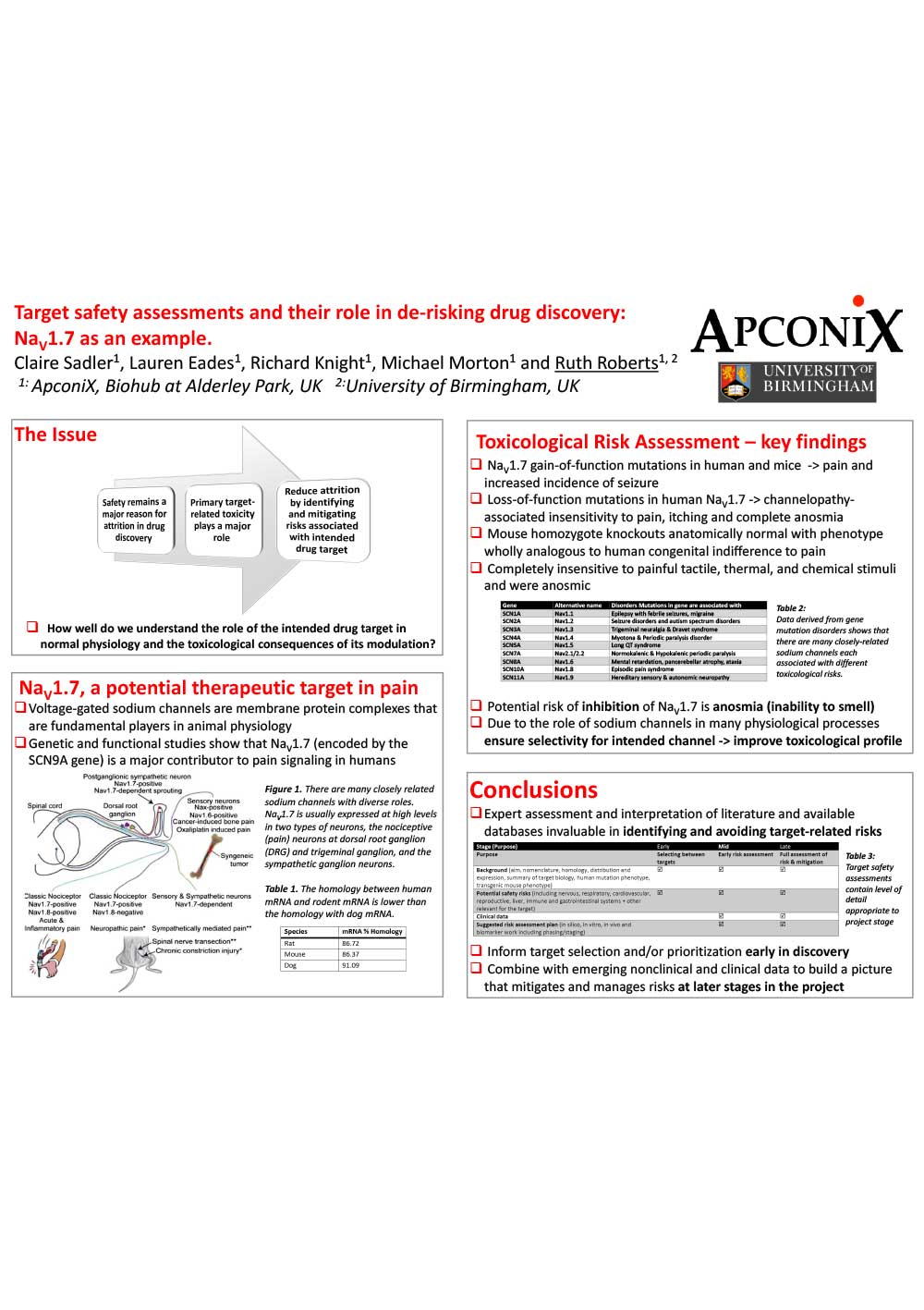
Target safety assessments and their role in de-risking drug discovery NaV1.7 as an example
[sdm_download id="10350" fancy="0"]

The CiPA Profile of Two Adenosine Uptake Inhibitors dilazep and dipyridamole
[sdm_download id="10351" fancy="0"]
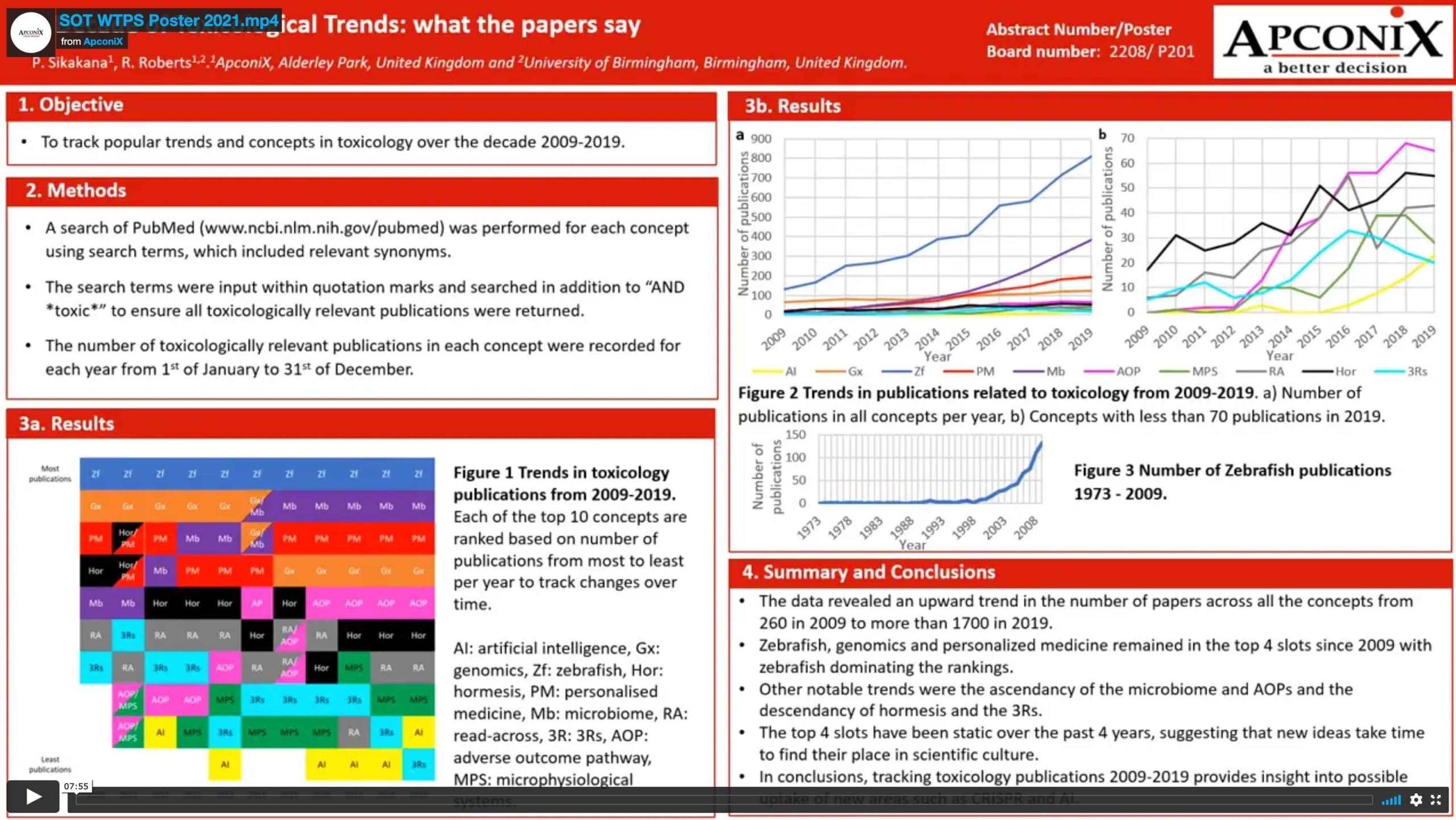
SOT WTPS to Poster Commentary 2021
[sdm_download id="10353" fancy="0"]