“Oral bioavailability in the rat is good, so we think this is a good clinical candidate”
I’ve heard variations on this comment a number of times over the course of my career in drug discovery projects.
But is it a true and useful statement?
Yes and no…I’ll try and explain.
Firstly, let’s take a look at the definition of oral bioavailability (or F for bioavailable Fraction):
‘Oral bioavailability is the fraction of the oral dose that is absorbed and escapes first pass extraction.’
Most commonly, first pass extraction is taken to mean removal by the liver, either metabolically (we have pretty good in vitro methods for establishing how rapid this rate is), or via biliary excretion (less so). It can of course also mean first pass extraction by the gut wall but for the sake of simplicity, let’s look at the circumstances where hepatic metabolism is the predominant source of first pass extraction.

Looking at the physiology that causes these effects, most (≥ 80%) of the blood supply leaving the gut, which therefore contains orally absorbed compounds (including drugs), drains into the Hepatic Portal Vein. It then travels directly to the liver, making it the first organ responsible for removal of absorbed compounds encountered by them (except the gut of course).
The liver is designed to be very efficient at removing foreign compounds, and this design enables it to remove absorbed compounds before they encounter any other part of the body, thereby minimising potential for toxicity.
Where F= bioavailability
Fabs is the fraction absorbed orally
Cl is total clearance for a particular species
Q is the liver blood flow
Cl/Q is the fraction of liver flow that total clearance represents
and Fabs [Cl/Q] is the fraction of the absorbed dose that is removed by the first pass hepatic extraction
This simplifies to:
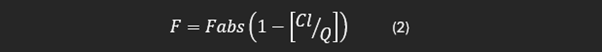
And rearranging equation (2), we can get to a means of calculating Fabs from bioavailability and clearance thus:

Now, Fabs is a very useful term (perhaps more so than bioavailability); by definition it describes that fraction of an oral dose that is absorbed. In most cases, to be orally absorbed a drug simply needs to dissolve in gut contents then diffuse across the membrane of the GI epithelium to the blood. It is therefore independent of clearance (which will vary as a fraction of liver blood flow across species), and one might think that it could be a means to predict human absorption from nonclinical data. If you thought that I’d very much agree with you!
However, despite the clearance independence associated with Fabs, to estimate it backwards from F we have to use both bioavailability and clearance in equation 3, which means that we need to include some assumptions, the most important of which is that clearance is predominantly hepatic.
It is worth noting here that the clearance we measure in an IV PK study is total clearance; that is, it is the sum of all clearance processes:
It shouldn’t be surprising that clearance isn’t always principally hepatic and therefore predictable from hepatocytes or microsomes, but it is a useful first assumption to make and is worth testing with as much rigour as possible (see below).
It is not uncommon to calculate Fabs from bioavailability data in rat or mouse, and find that it is either a very large number, i.e. much greater than 100% (implying that clearance approaches Q or in other words that Cl/Q approaches unity, unlikely if bioavailability is good I think?) or is negative, which means that Cl/Q>1, either of which are clear impossibilities if clearance is hepatic!
So, what am I saying here? Well, ‘good’ oral bioavailability (insert your own preferred percentage here) is a useful metric. However, to me Fabs is even more important; to predict bioavailability in humans (and the prediction of human properties of a drug is one of the principal aims of any Drug Discovery project), we need to have a reasonable idea of human and therefore preclinical Fabs.
And what would stop us from getting that point? Why, a reasonably thorough understanding of nonclinical and expected human clearance; if we don’t have that insight even sensible looking values in nonclinical species could be very misleading for the human picture.
Consequently, my view is: use bioavailability in mouse, rat or dog as a metric by all means, but ensure that you underpin it with a good understanding of the clearance of your compound/series.
Can you predict clearance in those species from hepatocyte and/or microsomal data? Do you know the rates of renal and biliary clearance? Do you know routes of metabolism?
If you do observe odd bioavailability data, have you looked at turnover in a gut system such as microsomes derived from enterocytes? Have you looked at transporter interactions?
Most of these assays are relatively cheap with rapid turnaround. There is no excuse not to have this data available; it is straightforward to profile your compound or series like this and will ultimately add lots of value to your asset.
My take-home messages:
- There is value in comparing oral bioavailability across a series.
- However, its reliability is dependent on the nature of clearance.
- To be confident in predicting human PK and in your bioavailability figures, you need to understand nonclinical and (as much as is feasible) human clearance.
- Doing this will increase your chances of finding a good clinical candidate!
