Analgesic Screening
ApconiX now offers screening of analgesic compounds in human iPSC sensory neurons.
The development of novel, non-addictive, therapeutics for the treatment of neuropathic pain is a global priority, as highlighted by the recent document released by the FDA (see button below). A way to circumnavigate addictive potential, is to specifically target peripheral sensory neurons, located within the dorsal root ganglia, which become sensitised in neuropathic pain states.
One of the key problems faced in pre-clinical efficacy testing is that findings in animal models often fail to translate to humans. An exciting alternative approach is to harness human induced pluripotent stem cell (hiPSC) technology, to assess the activity of novel peripherally acting therapeutics.
Given ApconiX’s globally established expertise in ion channels, iPSCs models and all-things electrophysiological we have successfully characterised functional expression of key ion channel targets in sensory iPSCs. We have also established a screening assay, using the multi-electrode array, which has shown to be sensitive to peripherally acting analgesics.
A collaboration with Medicines Discovery Catapult (MDC) incorporated calcium imaging, immunocytochemistry and biochemical assays and adding inter-laboratory comparison to our validation studies. MDC blog highlights some of the work here
Our hiPSC assay for screening peripheral analgesics
Our bespoke packages include MEA (whole-well or single electrode (16 per well) metrics and manual patch clamp.
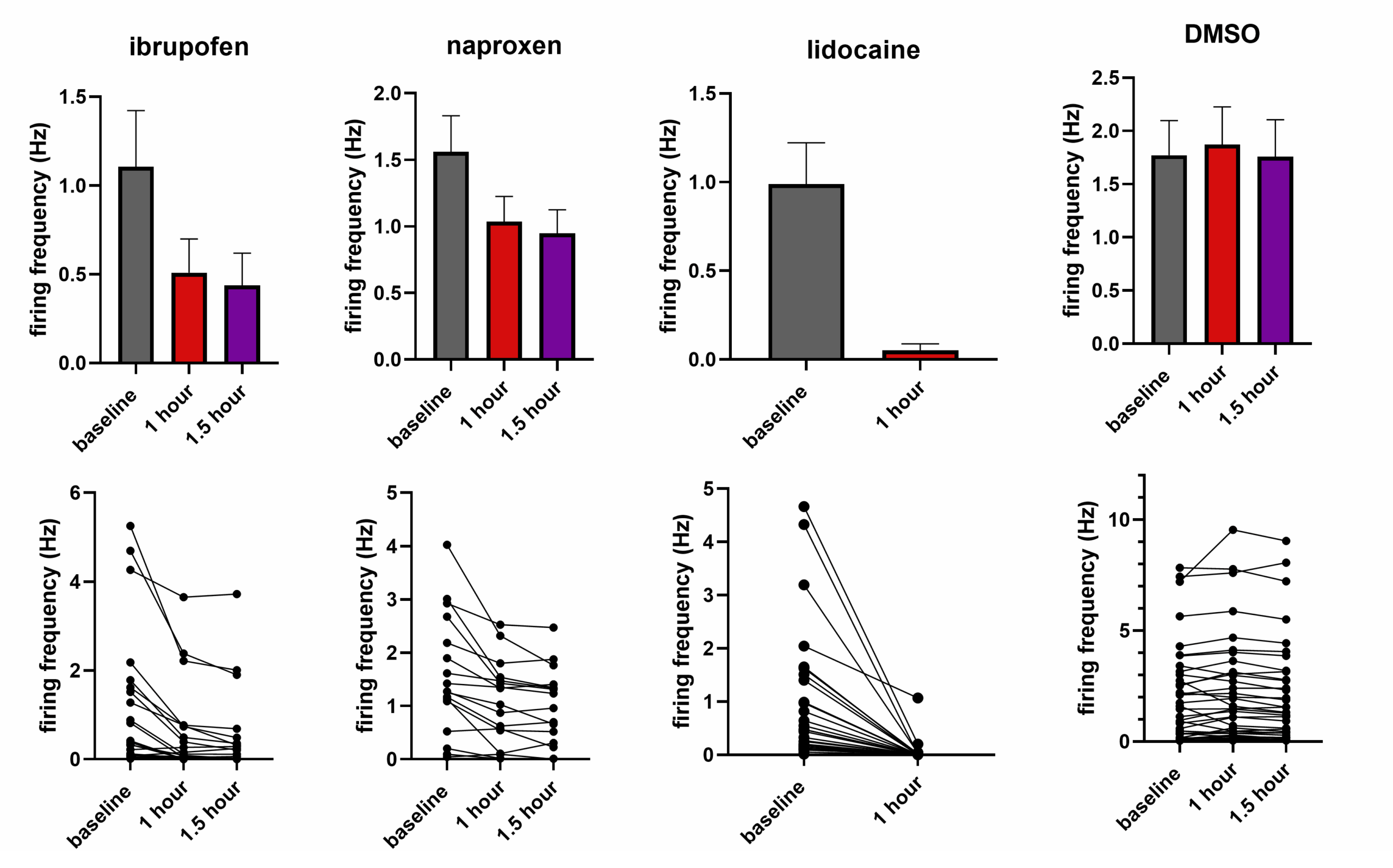
Summary bar charts showing effects of ibuprofen, naproxen (both 400µM), lidocaine (100µM) and DMSO (3:1000) on firing frequency.
We have characterized the functional expression of many key “pain” targets in RealDRG™ cells (see below). We show here that spontaneous activity in the cells could be modulated by established analgesics.
As shown left, incubation with ibuprofen, naproxen and lidocaine caused clear reductions in firing frequency, whilst DMSO was without effect.
A hot topic currently being explored in analgesic drug development is the use of cannabinoids to treat neuropathic pain. We compared the effects of the main psychoactive compound within cannabis, Δ9-tetrahydrocannabinol (THC) with a non-psychoactive central nervous system impermeant cannabinoid, cannabinol (CBN).
Both THC and CBN were shown to dramatically reduce firing frequency, whilst the methanol vehicle control was without effect, right. These findings indicate the potential use of isolated cannabinoids as novel analgesics, particularly CBN which specifically effects peripheral neurons. CBN would therefore circumvent any addictive potential or side-effects associated with psychoactive cannabinoids such as THC.
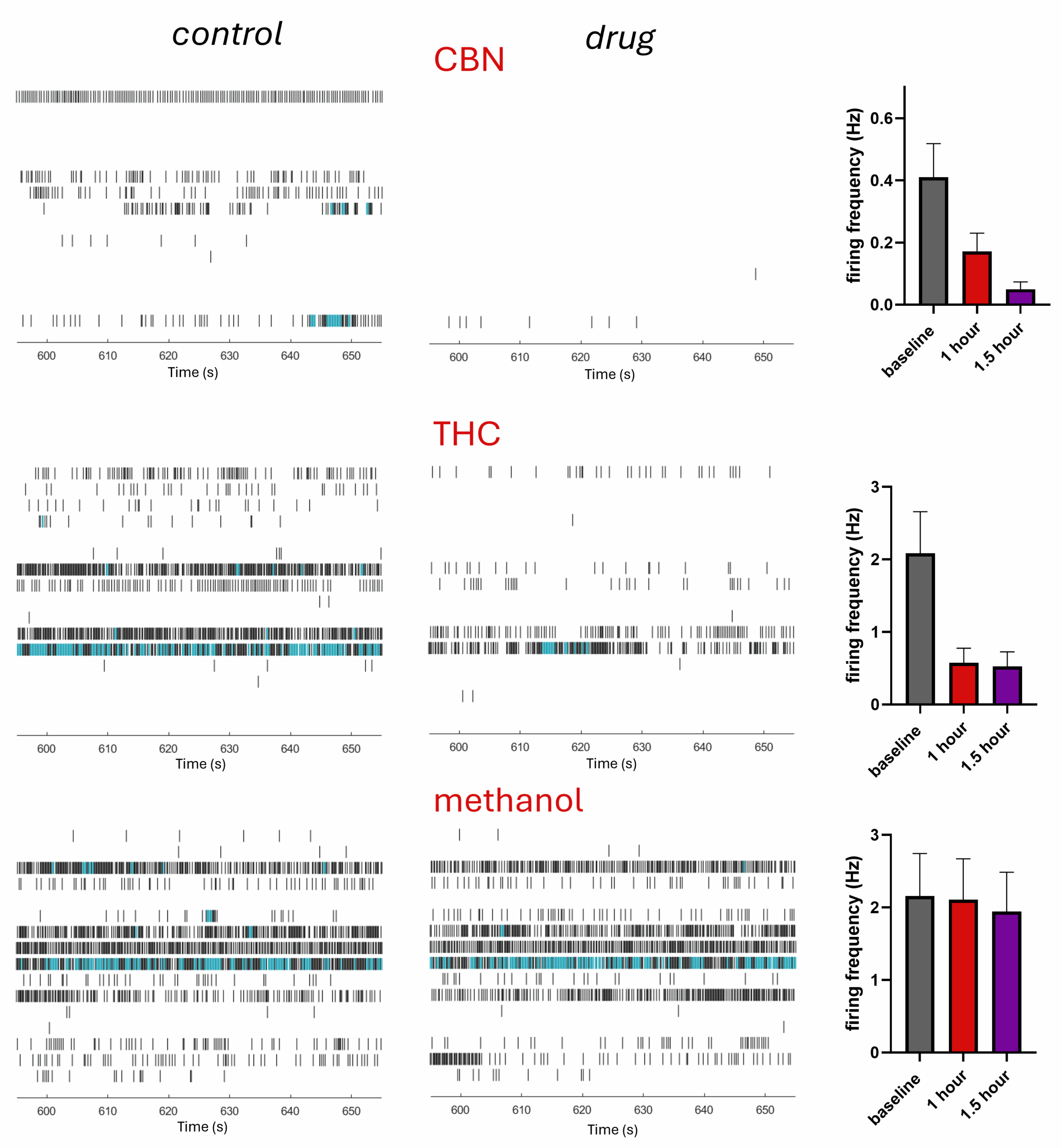
Example raster plots following 90mins application (LHS), and summary bar charts (RHS), showing effects of cannabinol (CBN), tetrahydrocannabinol (THC, both 9µM) and matched methanol control, following 60 and 90mins application.
Characterising key ionic mediators in RealDRG™
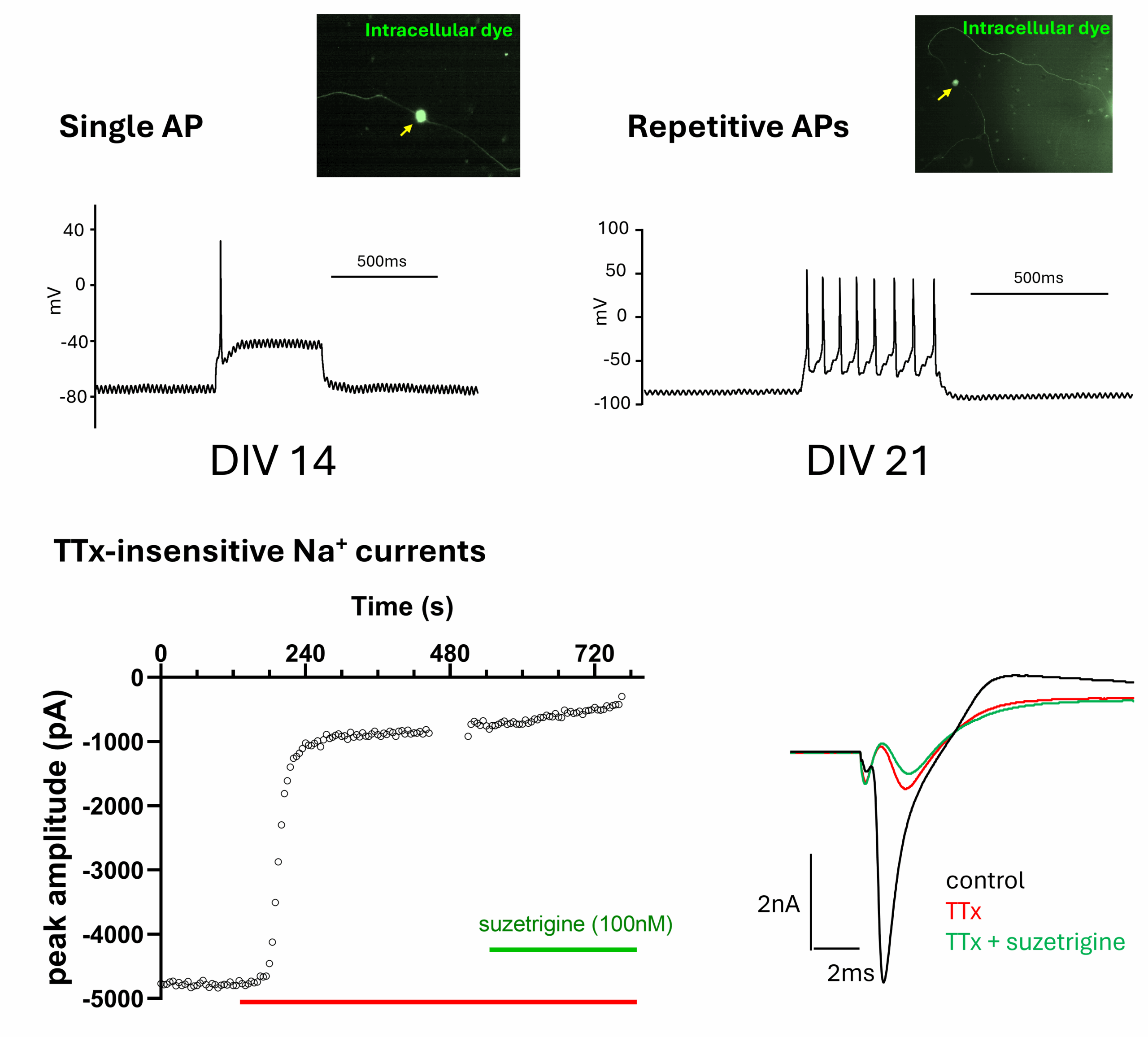
Top: example voltage traces showing single (LHS) and repetitive action potential firing (RHS) at days in vitro (DIV) 14 and 21 respectively. Inset images showing corresponding cell morphology as revealed by inclusion of fluorescent dye within recording solution. Bottom, example time course (LHS) and current trace (RHS) showing partial inhibition of peak Na+ current with TTx ± suzetrigine (both 100nM).
Spontaneous activity was rapidly abolished by TTx, indicating strong functional expression of NaV 1.7, however given that TTx-insensitive sodium (NaV1.8 and NaV1.9) channels, (common “pain” targets), are typically activated at higher membrane potentials we used manual patch clamp to examine their functional expression and contribution to sensory hiPSC excitability.
Manual patch-clamp recordings revealed a diversity in action potential (AP) waveform and maximal firing frequency in response to current injections. Inclusion of a fluorescent tracer within the patch-pipette enabled us to visualise the characteristic unipolar morphology of sensory neurons. Sensory hiPSCs typically responded to 500ms stimulation with a single AP, however with extended time in culture (>DIV20) a proportion of neurons exhibited repetitive AP trains, right. This marker of neuronal phenotypic maturity was accompanied by the emergence of TTx-insensitive sodium currents in some cells, which were on occasion sensitive to suzetrigine, specifically implicating functional NaV 1.8 expression, left.
Collectively these observations reflect the functional heterogeneity of sensory hiPSCs and closely mirror features of ex vivo human DRG neurons, and the in vivo setting.
Key “pain” targets are expressed in RealDRG™ cells
Real-time application of various agonists to RealDRGTM (Anatomic, MA, USA) could evoke changes in the frequency of LFP spikes and enabled us to quantify the percentage of sensory hiPSCs functionally expressing a given target. As exemplified right, increases in firing frequency were commonly observed in response to α-β-Me-ATP and histamine whilst the matched vehicle control (media) was without effect. This showed that 86% of cells functionally expressed P2X2/3 receptors whilst 30% expressed Hrh1-4 receptors. A smaller proportion of cells (26%) also responded to capsaicin, verifying functional TRPV1 in a subset of RealDRGTM. Notably, 19% of cells showed responsivity to DMSO, however these responses were of smaller amplitude and qualitatively dissimilar to capsaicin.
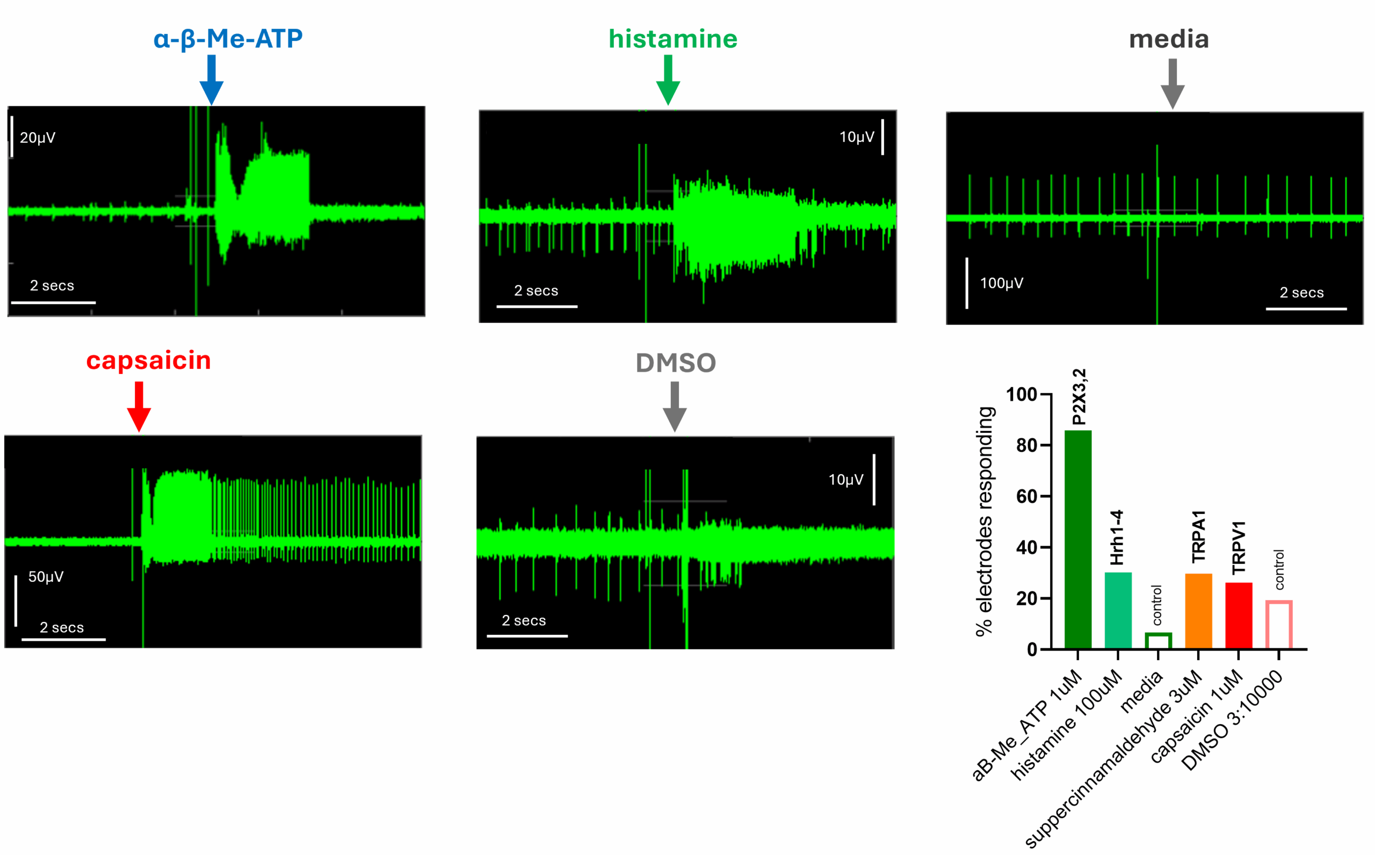
Example MEA traces showing responses to α-β-Me-ATP (1µM), histamine (100µM), media control, capsaicin (1µM) and DMSO (3:10000). Bottom RHS, bar chart showing % of cells responding to each agonist.
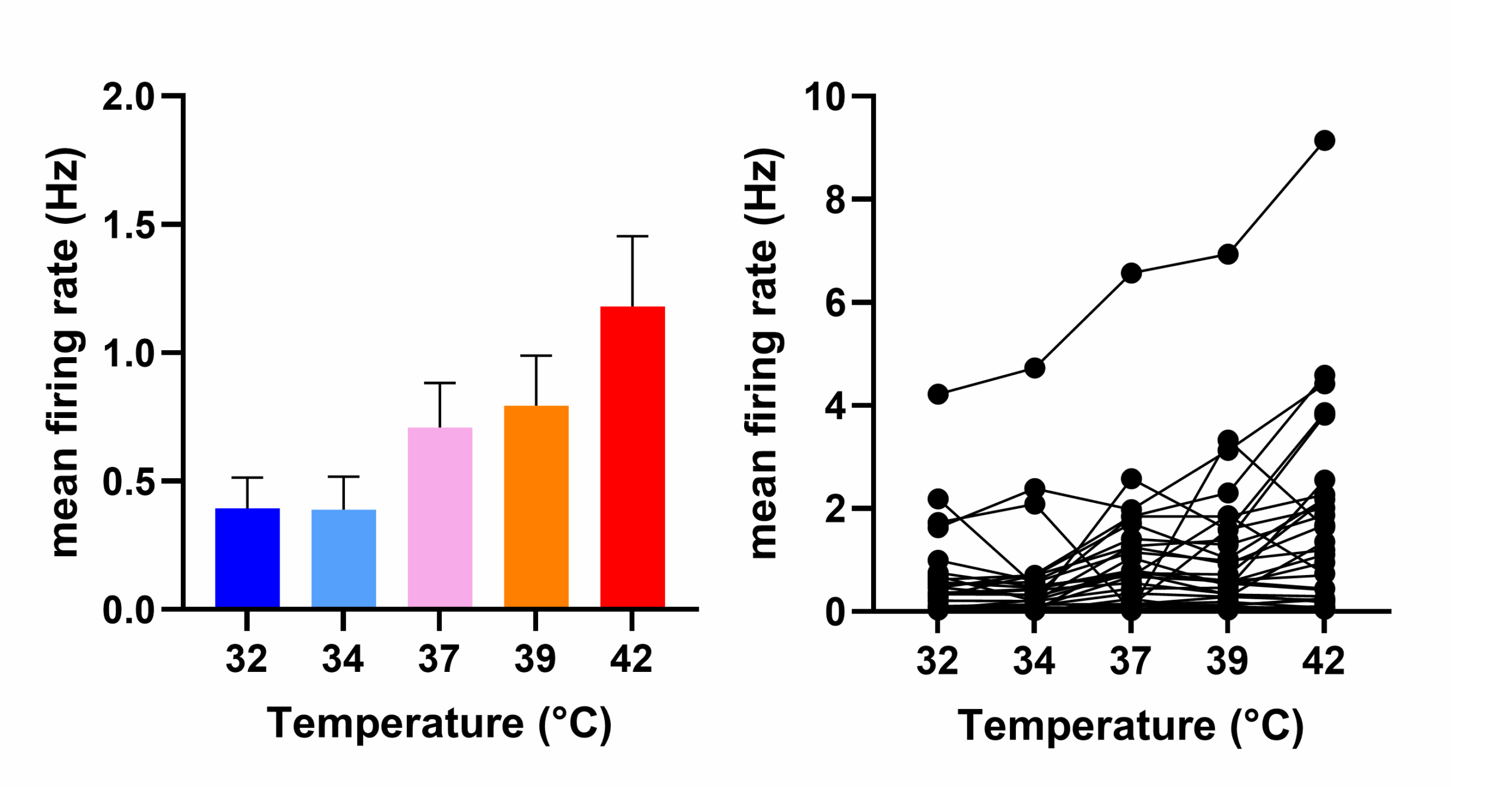
Bar charts showing a subset of sensory hiPSCs exhibiting elevated firing frequencies in response to increasing temperature.
MEA recordings revealed many RealDRGTM exhibited spontaneous LFPs, resembling the increase in spontaneous discharges recorded from human sensory neurons in neuropathic pain patients. A subset of hiPSCs responded to temperature elevations with increased firing frequency, a key feature of some sensory neurons.



